Where to Start?
Start With Vacuum Purging the Test Item
Across various industries, vacuum purging is used to remove volatile substances/chemicals from the test item at low temperatures first before heating. Under vacuum, butane, propane, and other solvents convert into a gaseous form and evaporate, also known as “outgassing.” Vacuum purging works by lowering the atmospheric pressure in a vacuum oven chamber. Atmospheric pressure prevents “outgassing” from the surface area of the test specimen.
Outgassing is a concern for any material intended for use in high-vacuum environments. It refers to the release of gas trapped within a solid. As the pressure decreases in the oven, the volatility increases allowing chemicals to boil off at lower temperatures without heating the oven. Contamination of surfaces is a source of outgassing. After production, the surface of vacuum components is always contaminated.
They must be thoroughly cleaned before installation.
The vacuum drying oven has the advantages of fast drying of specific items, little pollution, and no damage to the inherent quality of the dried items. But, test specimens placed in vacuum ovens should be evacuated before heat is used.
Some reasons are listed below:
- The test specimen is put into a vacuum oven and is evacuated to remove the gas from the material. If the work piece is heated first, the gas will expand when heated. This can result in high pressure generated by the expanding gases enough to overcome the capacity of the vacuum pump. Under certain conditions, excessive pressure may cause the tempered glass observation window to burst. We can avoid this issue when we provide vacuum environment first and then heat later.
- If you heat first and then evacuate, the heat will be transferred to the vacuum pump when the hot gases are evacuated. This situation can cause the temperature of the vacuum pump to rise too high and reduce the efficiency of the pump.
- The heated gases can affect the vacuum pressure gauge as temperatures rise. This situation may cause the display error on the vacuum pressure gauge.
Remember, the correct method is to evacuate first and then heat. If time permits, raise the temperature slowly until the desire temperature is reached. Water vapor is typically removed at bakeout temp of at least 120°C or 250°F for 12 hours.
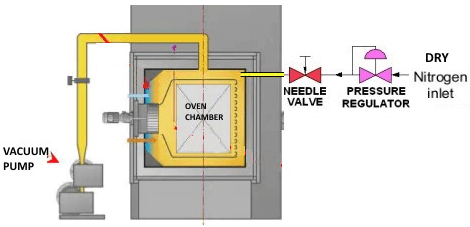
Venting with dry N2 gas forms a nitrogen blanket into the oven chamber surface. As the dry N2 gas sweeps across the reservoir, it will absorb water vapor which is forced out of material as it moves towards moisture equilibrium with the nitrogen blanket.
Nitrogen is generally a reliable dry gas, which is economic and safe. This Nitrogen vent saves reintroducing contaminates and water vapor into the oven chamber during the next run.
For more information or help with your vacuum applications:
Contact: carl@mackvacuum.com
